- Subscribe
- Digital Edition
-
Beverage Guide
- Flavor and Cocktail Construction >
- Production Fundamentals >
- Non-Alcoholic Beverages >
-
Beer
>
- Cider >
- Sake
-
Spirits
>
-
Wine
>
- Kamaʻāina Wine Makers >
-
Winemaking
>
- A Guide to: High Sugar Residual Wines
- A Guide to: Post Fermentation Flavor Adjustments
- A Guide to: Post Fermentation Process: Stabilization
- A Guide to: Wine Prefermentation Practices
- A Guide to: Wine Microbes
- A Guide to: Wine Alcoholic Fermentation Physical Environment
- A Guide to: Wine Fermentation Chemical Environment
- A Guide to: Wine Bottling
- A Guide to: Wine Faults
- A Guide to: Wine Polyphenols
- A Guide to: Wine Aroma Compounds: Pt 1
- A Guide to: Wine Aroma Compounds: Pt 2
- A Guide to Viticulture
- Red and White Grape Aroma Compounds
- Wine Styles >
- Business Strategy
- News and Events
- About
- Production Fundamentals
- Flower Aroma Compounds
*This article is a compendium to alcohol removal by physical methodologies which can be found here: hawaiibevguide.com/alcohol-removal
For the production of non-alcoholic beer and wine, natural fermentation techniques can be used to reduce or eliminate the formation of ethanol. These techniques require less up-front cost and no special equipment, but instead they use what’s already on hand.
For the production of non-alcoholic beer and wine, natural fermentation techniques can be used to reduce or eliminate the formation of ethanol. These techniques require less up-front cost and no special equipment, but instead they use what’s already on hand.
Good Overview articles
|
[1] Brányik, T., Silva, D. P., Baszczyňski, M., Lehnert, R., & e Silva, J. B. A. (2012). A review of methods of low alcohol and alcohol-free beer production. Journal of Food Engineering, 108(4), 493-506. Retrieved from: https://pdfslide.net/
documents/a-review-of-methods-of-low-alcohol-and-alcohol-free-beer-production.html?page=1 [2] Montanari, L., Marconi, O., Mayer, H., & Fantozzi, P. (2009). Production of alcohol-free beer. In Beer in health and disease prevention (pp. 61-75). Academic Press. Retrieved from: https://www.researchgate.net/ publication/286327980_Production_of_alcohol-free_beer [3] Piornos, J. A., Koussissi, E., Balagiannis, D. P., Brouwer, E., & Parker, J. K. (2022). Alcohol-free and low-alcohol beers: Aroma chemistry and sensory characteristics. Comprehensive Reviews in Food Science and Food Safety. https://ift.onlinelibrary.wiley.com/doi/full/10.1111/1541-4337.13068 Brewing Terms to know Attenuation The degree to which yeast ferments the sugar in a wort or must. For example, 50% attenuation means that 50% of the sugars have been converted into alcohol and CO2 by the yeast. Likewise, 100% attenuation means all of the sugars have been consumed by yeast. Yeast pitching The process of adding yeast to wort to start fermentation [4]. Aliphatic alcohol Alcohols with an open chain rather than a ring structure (aromatic) [5]. Original gravity (original extract) [6] A measure of the original (before alcoholic fermentation) concentration of solids in the wort, expressed as the density above that of distilled water at standard temperature and pressure (Abbreviated “STP” this is typically 20 °C/68 °F and 760 mm Hg). For example: Water’s density at STP is 1.000. A wort density of 1.048 means it will have 48° of excess gravity and an OG of 48. • Original gravity can be used to determine a beer’s alcohol potential and is measured in the brewery by saccharometer, hydrometer, or refractometer. |
• While original gravity is used in brewing, alternative measurements for concentration of solids in wort can be expressed as degrees Plato, degrees Balling, or percent dry matter of the wort (% wt.), and Brix. To calculate this difference between OG and degrees of dissolved solids (Brix for example), the units are approximately one-quarter of the excess gravity. For example, an OG of 1.048 can be converted to °Brix by 48/4 = 12° Brix/Plato. Typical wort gravity [7]:
• Normal gravity wort: Typically 1.040-1.052 (10°–13° Plato) typically results in a beer containing 4%–6% alcohol by volume (ABV). • High gravity wort: Typically 1.056-1.068 (14°–17°Plato) will result in a beer of 6%–8% ABV. |
Modified Mashing Regime
In the brewing of alcoholic beer, the mashing process involves mixing the milled grain with warm water. This is done to break down complex molecules like starch into sugar, by activating amylase enzymes and proteases, which break down proteins. To formulate a mash, the temperature of the water is used to adjust the rate of these enzyme actions to manipulate the resulting sugars. In the brewing of non-alcoholic beer, the mashing regime can be changed to limit the amount of starch converted into fermentable sugars, thereby reducing the amount of ethanol that can form [8]. In particular, the spectrum of sugars formed depends on the actual enzyme activities present [1].
|
Mechanisms
Temperature influences enzyme activity and can be manipulated to control the carbohydrate composition and the fermentability of the final wort. α- amylases Mechanisms Converts starch first into non-fermentable sugars (dextrins) and, during prolonged action, into fermentable sugars of glucose, maltose, and maltotriose [2]. Higher activity results in more fermentable sugar. Optimal Temperature for function: • 65–70 °C [9] • 72–75 °C generates [1] Deactivation Temperature: 76–78 °C β-amylases Mechanisms Converts the longer polysaccharide segments like dextrins and soluble starch produced by α- amylase into maltose [1] [2] [3]. Optimal Temperature for function: 60–65 °C [1] Deactivation Temperature: While 76–78 °C can have some impact, β-amylase is more thermally stable at high temperatures than α-amylase. Process The temperature and process of mashing can be modified to deactivate enzymes. This can be done in various ways. Malt Type A percentage of dark or pale caramel malts in the mash can help to mask the wort flavor of arrested fermentation beer. Branyik (2012) notes that pale caramel malt contributes the highest amount of desirable Maillard reaction compounds like furfural and 2-acetylpyrrole. High Temperature Mashing [1] This process uses a generally consistant mash temperature to deactivate β-amylase where: • Mash temperature: 75–80 °C • Deactivates β-amylase while allowing a sufficient concentration of active α-amylases to digest and liquify the starch. Decoction Mashing [2] Mash decoction is the process of removing a part of the mash, boiling it to 215 °F (102 °C), then reintegrating it back into the main mash. This denatures enzymes and increases Maillard reaction products that provide the resulting beer worty aromas and darker colors[10]. Procedurally, for non-alcoholic beer: |
• The grist is briefly mashed with 52 °C (125 °F) water to activate some enzymes.
• The first decoction is performed. This consists of removing a percentage of the wort (decoction), which varies by the producer. • Subsequent decoctions may occur. The number of decoctions varies by producer and influences the concentration of Maillard reaction products, as the degree to which they are produced is influenced by how intensely the decoctions are boiled and the levels of protein modification (and hence the availability of amino acids) in the malt. For more on Maillard reactions, visit hawaiibevguide.com/maillard-reaction. • After reintegrating the decoction into the mash, the temperature and subsequent steps of lautering and sparging are conducted at or above 76 °C (168 °F) in order to further deactivate the amylases (mainly β-amylase) and prevent the formation of normal concentrations of fermentable sugars. • Wort acidification, by the addition of 5% acid malt or lactic acid 10 minutes before the end of boiling to a pH of 4.3, may be required to achieve a sufficient pH for low-alcohol beers. This because wort attenuation of ~10% creates a pH of ~5.0 and results in low liveliness,emphasizing the beer’s worty flavor. Final Wort for Low-Alcohol Beer • 25–30% fermentability (typically 80% fermentability for traditional pale beers). • Ideal original gravity: 4.0-7.5 wt.% [1] • Brewing at high gravity (20 wt.%) increases the formation of higher alcohols and esters. This phenomenon can be exploited to strengthen the flavor of reduced alcohol beers obtained after dilution of a higher gravity beer. Wort Dilution • Dilution from 11.5 to 7.5 wt.% original gravity after, rather than before, wort boiling resulted in lower bitterness, increased ester and higher alcohol levels and decreased the worty smell and taste. • From a taste perspective, Hawaii Beverage Guide has noticed that dilution may be a significant technique used in commercially available non-alcoholic beer produced by arrested fermentation. We have not been able to verify this technique, however. Hop Addition • Saerens and Swiegers (2014) recommend hop additions to be made at least 30 minutes before the end of boiling of the wort, and no dry hopping to prevent unintentional inoculation of the fermentation. • The ”Hop creep” refers to the ability for hop-produced enzymes to break down dextrins and cause re-fermentation. Hop oils can be used to mitigate this issue [12] |
Changes to Fermentation Process
|
Upon completion of the modified mashing procedure, low alcohol beers typically require adjustments to the fermentation process in order to restrict the formation of ethanol and develop desirable aroma characteristics.
Arrested Fermentation/Production Conceptually, arrested fermentation lets yeast metabolize normally, thereby producing desirable aroma compounds, and then stops the process before the ABV reaches .05%. While Montari et al. (2009) distinguishes between stopped fermentation, which is the removal of yeast before full attenuation, and limited fermentation, in which yeast metabolism is restrained, the distinguishing factors are too blurred for a consistent label for each process. Instead, specific fermentation parameters of time, temperature, and wort sugar levels are helpful. Branyik et al (2012) noted the following fermentation parameters: Temperature and time The temperature of fermentation’s initial phase can vary without a significant impact on the formation of volatiles and the reduction of aldehydes [1]. Example time and temperatures for <.5% ABV beer include: • 0-12 °C for 7 hours [1]. • 15–20 °C for 0.5–8 h [2]. • Higher temperatures increase ester and diacetyl formation, especially by Saccharomyces pastorianus (lager/bottom fermenting yeast) and decreased aldehydes. These accelerated reaction kinetics, however, require stopping fermentation shortly after the wort is pitched because of the increase in ethanol production. Oxygen The wort is not aerated during fermentation. While alcoholic fermentation is an anaerobic process, this lack of oxygen, according to Montanari et al (2009): Reduces yeast reproduction. • Lengthens the lag phase of yeast development, which is when the yeast consumes and metabolizes, but does not propagate or produce ethanol. • Increases the ester formation by Saccharomyces cerevisiae. • Like in wine, too little oxygen can result in reductive off-odors caused by sulfur compounds like dimethyl sulfide (DMS). And unlike wine, where micro-oxygenation can be used, it may need to be reduced after fermentation. Yeast Type • Saccharomyces cerevisiae (ale/top fermenting yeast) compared to Saccharomyces pastorianus (lager/bottom-fermenting yeast), form higher concentrations of higher alcohols and esters at lower attenuation [1], but also produce higher diacetyl content [2]. • Saccharomyces pastorianus (lager/bottom fermenting yeast) works better with a higher fermentation temperature as, despite the short fermentation time, the diacetyl level is reduced, compared to the same temperature used for top-fermenting yeasts [2]. • ~0 °C at Saccharomyces cerevisiae (ale/top fermenting yeast) has lower attenuation than for Saccharomyces pastorianus [2]. This is consistent with optimal lager fermentation temperatures being 45–55°F (7–13°C) where as ale |
fermentation temperatures are typically 60–75°F (16–24°C) [13].
• Alternative yeast can be used. See section: “Alternative Yeast” Stopping Fermentation The challenge in this process is to stop fermentation before ethanol is produced at too high a rate. For this reason the fermentation is constantly monitored. To arrest the fermentation, the beer is quickly cooled to 0 °C and the yeast is removed by centrifugation or filtration [1]. Cold Contact Fermentation (Cold Contact Process) Cold contact fermentation (CCF) is a specific type of arrested fermentation in which low temperature is used to slow ethanol production by limiting yeast metabolism, while higher alcohols and esters form and carbonyls and aldehydes that are suspected of imparting worty flavor are reduced [1] [2]. Fermentation Parameters [1] Wort: • 6 wt.%. • Wort may be cooled to 0–1 °C before pitching. Temperature and duration • 0–1 °C and ideally −0.5 to −0.4 °C for 49 hours [3]. • 2–3 °C for 150–200 hours [2]. pH: Acidified with lactic acid to pH 4. Yeast cell concentration 30x10^6 cells/ml. However, when using a high yeast cell concentration (>30x10*8 cells/ml), the yeast slurry used for pitching already has an ethanol content of ~6 ABV. Cold contact with immobilized yeast An experimental process utilizes cold contact fermentation with immobilized yeast in-lieu of batch cold contact fermentation as the latter is a slow and difficult process to control. The use of immobilized yeast expedites the process, but immobilizing the yeast is difficult and requires special equipment [2]. Post Fermentation Treatment The beer is carbonated, stabilized, and sterilized. In this set of processes, Brányik et al (2012) notes: Microbial stabilization to eliminate unwanted fermentation can be done by pasteurization or filtration. For more on microbial stabilization by filtration: www.hawaiibevguide.com/filtration.html Undesirable aroma reduction from carbonyls, DMS and other sulfur compounds, low-alcohol beer may then be: • Matured for at least 10 days at 0 to 1 °C [1]. • Cooled to ~20 °C to avoid foam production, then sparged under pressure with nitrogen (N2) or carbon dioxide (CO2) to eliminate any sulfur compounds that were not evaporated during wort boiling [2]. This is critical, because while yeasts typically eliminate them during normal fermentation, yeast metabolism during CCF is limited. Montanari et al (2009) notes that CO2 is preferable because of its presence in the final beer at a concentration of 5-6 g/L. CO2 prevents spontaneous fermentation. Ascorbic acid can be added to hot wort to minimize the unpleasant bitter aftertaste of oxidized malt substances [1]. |
Alternative yeast
|
Mechanism
Alternative microbes produce limited quantities of ethanol because they cannot effectively ferment sugars in wort while ideally also producing large amounts of flavor-active compounds during fermentation. The specific reasons as to why the yeast cannot ferment the sugars varies by species used. Strejc et al., (2013) notes that mechanisms can include gene deletions in the tricarboxylic-acid cycle [14], and gene modifications which influence enzyme expression like the elimination of alcohol dehydrogenase, or for increased glycerol production, and lager strains with overexpressed glycerol-3-phosphate dehydrogenase [15]. Challenges of using alternative yeast • Strains are usually proprietary and not re-pitchable. • The development of new strains of microorganisms for the brewing industry might raise concerns for both producers and consumers, because of the uncertainty that this could generate from the consumers’ perspective [16]. • When using alternative yeast, it is important to keep the wort and the beer free from other potential microbes (Saerens and Swiegers 2014). Additionally, since a lot of sugar is left in the final beer and there is no ethanol to act as a preservative, extra care should be taken to avoid spoilage. • Yeast may or may not be hop-tolerant, which can be a challenge for styles like IPAs. This can be mitigated by post fermentation hop addition using hop oils given the necessary sterile environment as noted by Saerens and Swiegers (2014). Species Types While there are a multitude of experimental yeasts, the following are those that are commercially available. Pichia kluyveri Commercial example: CHR Hansen NEER • Fermentation Temperature: 16-22 °C (59-71 °F) with an optimal temperature of 18 °C (65 °F). • Fermentation duration: 2-6 days • CHR Hansen Notes: Isolated in New Zealand, this strain was chosen for its enhanced fruit flavors produced by esters and thiols, no production of diacetyl allowing for shortened maturation cycles, lower volatile acidity, and rounder mouth-feel, as well as a medium production of polysaccharides for “next-generation, full-flavored, yet fresh, alcohol-free beer”. Additionally, when given oxygen, it will respire and be metabolically active without producing any ethanol. • www.chr-hansen.com/en/food-cultures-and-enzymes/ femented-beverages/cards/product-cards/neer Mechanisms Saerens and Swiegers (2014), of CHR Hansen, in a patent application, note that Pichia kluyveri yeast is only capable of fermenting the glucose, therefore an ideal concentration of glucose in wort is 0.01-3% (w/vol). When compared to commercial pilsner beers like Carsberg, Stella, and Heineken, decreased concentrations of ethyl butyrate and ethyl hexanoate, increased or similar concentrations of isoamyl acetate and isoamyl alcohol, and similar concentrations of ethyl octanoate were found. |
Hanseniaspora uvarum
Commercial Example: Escarpment Laboratories NAY • Temperature: 20-25 ºC (68-77 ºF) • Attenuation: 10-20% • Notes from Escarpment Laboratories: • “Whether you want a clean, refreshing lager or a fruitier, hoppier IPA, NAY will help you achieve the perfect results. Scientifically, it is a strain of Hanseniaspora uvarum yeast we isolated from spontaneously fermenting crabapples here in Guelph (Ontario, Canada). NAY is a maltose-negative yeast, meaning it does not ferment most of the sugars in wort. It is non-phenolic and non-diastatic.” For additional insight into Escarpment Laboratories’ yeast selection process: https://escarpmentlabs.com/blogs/ resources/approaches-to-non-alcoholic-beer-fermentation Saccharomyces cerevisiae var. chevalieri Commercial example: Fermentis by Lesaffre SafBrew LA-01 • Fermentation Temperature: 15-25 °C (59-77 °F). • Notes from Fermentis: “This yeast does not assimilate maltose and maltotriose but assimilates simple sugars (glucose, fructose and sucrose) and is characterized by a subtle aroma profile.” • https://fermentis.com/en/product/safbrew-la-01/ Saccharomycodes ludwigii Commercial example: White Labs WLP618 • Fermentation temperature: 68-75 °F (20-24 °C) • White Labs notes: “This specific strain has been chosen because of its lower ethyl acetate production compared to similar strains.” • www.whitelabs.com/yeast-single?id=179&type=YEAST Mechanisms Saccharomycodes ludwigii cannot ferment maltose because it does not have invertase and maltase [2]. By changing mashing techniques to limit glucose and sucrose sugars, ethanol production can be reduced [17]. Torulaspora delbrueckii Commercial example: White Labs WLP603 • Fermentation temperature: 66- 74 °F (19-23 °C) • White Labs Notes: “The profile of this strain has high ester production and will lend well to styles such as a fruit-forward IPA or Saison.” Additionally, “this particular strain was isolated from apples from an island in Denmark.” • www.whitelabs.com/yeast-single?id=176&type=YEAST Mechanisms Torulaspora delbrueckii is more capable of the removal of wort-derived aldehyde off-flavors than S. ludwigii because of its adequate aldehyde reductase activity, and is sufficiently sensitive to low temperatures to limit the formation of ethanol. For more insight: Nikulin, J., Aisala, H., & Gibson, B. (2022). Production of non-alcoholic beer via cold contact fermentation with Torulaspora delbrueckii. Journal of the Institute of Brewing, 128(1), 28-35. https://onlinelibrary.wiley.com/doi/full/10.1002/jib.681 |
Impact of Incomplete Fermentation
|
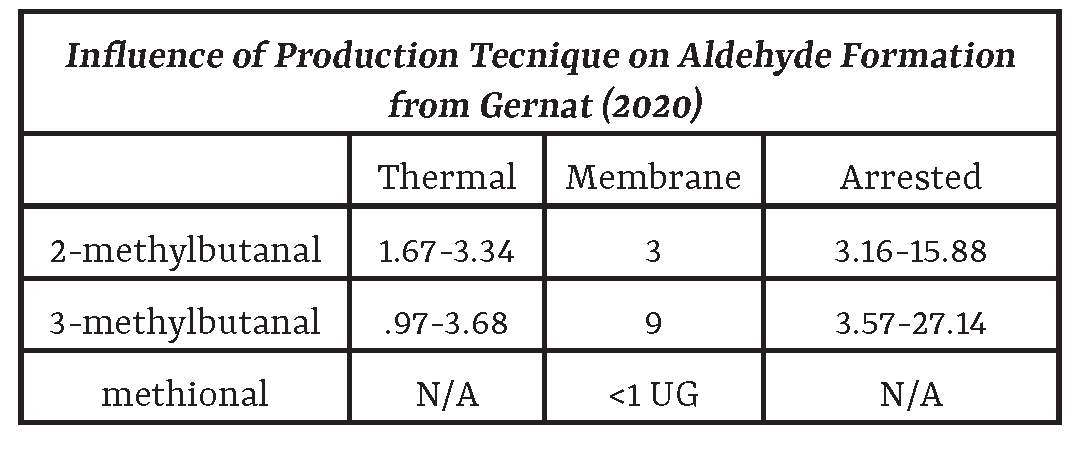
Aldehydes
Gernat et al. (2020), in a literature review, provided the following insight into aldehyde formation, as well as its influence on non-alcoholic beer [20]. We highly recommend reading the full article for an in-depth understanding of the process. Formation Aldehydes can form through a multitude of chemical pathways during brewing, and typically involve the usage of heat. These processes include: • Maillard reaction and Strecker degradation during processes that involve the application of heat, including malt kilning, mashing, and wort boiling. For more on Maillard reactions, read: hawaiibevguide.com/maillard-reaction • Oxidation of unsaturated fatty acids by enzymatic oxidation, autoxidation and photo-oxidation • Degradation of bitter acids • Aldol condensation • Melanoid-catalyzed oxidation of higher alcohols • Secondary autoxidation of aldehydes • Aldehyde secretion by fermenting yeast Impact on Beer In alcoholic beer, aldehydes are the primary contributors to the malty, worty aroma. In non-alcoholic beer: • Due to the reduction of ethanol, higher alcohols, and esters, the worty, malty aroma can be out of balance, and therefore excessive. • When non-alcoholic beer is produced by biological methods, higher concentrations of aldehydes can occur due to the incomplete conversion into higher alcohols. For more on this conversion, see the following section “Higher Alcohols.” It should be noted that this conversion occurs for dealcoholized beers, as they are completely fermented. • The absence of ethanol increases the polarity of the beverage matrix and results in an increased partition coefficient for more non-polar aldehydes. This, in turn, increases aldehyde volatility, which results in an increase in worty character because the molecules of volatile aroma compounds which are actually volatilized (in vapor phase as opposed to liquid phase) are what is perceived as aroma. Brányik et al (2012) notes the following change: • In regular beer, aldehyde retention: 32-39% • In non-alcoholic (0.5% ABV) beer: 8-12% retention • Monosaccharide’s higher concentration in non-alcoholic beers, compared to alcohol containing beer, can also reduce aldehyde’s solubility, and therefore increases volatility in beer. • The synergic interaction of aldehydes like 2-methylbutanal and 3-methylbutanal to sulfur-containing degradation products of methional may enhance the unpleasant taste of non-alcoholic beers. |
Important types in non-alcoholic beer:
Strecker aldehydes are the most significant contributor to malty and worty aromas, and have very low perception thresholds [20]. The most influential are 3-methylbutanal (malty), methional (cooked potato. 2-methylbutanal (malty aroma) may be significant, but was only confirmed by (Perpète & Collin, (1999) [21]. Methylpropanal (malty) may also be significant [16]. Common aldehydes in beer include acetaldehyde (green, vegetable) [22], hexanal (green, grassy), (E)-2-nonenal, benzaldehyde (cherry, almond), furfural (malty), and 5-hydroxymethyl-furfural (malty), phenylacetaldehyde (honey, malty). Influenced By The primary reduction mechanism of aldehydes during fermentation is the Ehrlich Pathway in which aldehydes are transformed into higher alcohols or fatty acids depending on the redox status of the yeast. For more see section “Higher Alcohols” Zygosaccharomyces lentus Commercial example: White Labs WLP686 • Fermentation Temperature: 68-75 °F (20-24 °C) • White Labs Notes: The profile of this strain is found to be very neutral. This species is slower and might take longer to reduce sugars and lower the pH of the beer compared to other strains. Little ester production was perceived in lab-scale trials. • www.whitelabs.com/yeast-single?id=197&type=YEAST Other yeast Other strains that have potential to produce non-alcoholic beer, which are noted by Piornos et al (2022) and Muller et al (2019) but do not appear to be available commercially are: • Saccharomyces rouxii spp. [18] • Spontaneous mutants of Saccharomyces pastorianus • Cyberlindnera spp. • Lachancea fermentati [19] For additional insight into yeast: Strejc, J.; Siříšťová, L.; Karabín, M.; Almeida e Silva, J.B.; Brányik, T. Production of Alcohol-Free Beer with Elevated Amounts of Flavouring Compounds Using Lager Yeast Mutants. J. Inst. Brew. 2013, 119, 149–155. https://doi.org/10.1002/jib.72 Bellut, Konstantin & Arendt, Elke. (2019). Chance and Challenge: Non- Saccharomyces Yeasts in Nonalcoholic and Low Alcohol Beer Brewing – A Review. Journal of the American Society of Brewing Chemists. 77. 1-15. 10.1080/03610470.2019.1569452. Retrieved from: /www.researchgate.net/publication/331943504_Chance_and_Challenge_Non-_Saccharomyces_ Yeasts_in_ Nonalcoholic_and_Low_Alcohol_Beer_Brewing_-_A_Review |
|
Reduction of aldehydes during cold contact fermentations
•While yeasts that are heat-deactivated so that they no longer produce ethanol can provide limited aldehyde reduction, active yeasts are required for larger conversions of both linear and branched aldehydes like those produced by Strecker degradation. However, Perpète and Collin (1999) [23] did not clearly conclude if aldehyde concentration reduction is enzymatic or adsorptive. While noting heat inactivation was at 60 °C for 20 minutes, to the extent that the cells were unable to grow, they did not distinguish between intact but metabolic inactive cells, and degraded yeast cells [23]. • It should be noted that yeast, during CCF, can produce aldehydes like 3-MB. The rate is slow, and can be mitigated by close monitoring. • The key yeast-produced enzymes in this reduction process are aldehyde dehydrogenase and aldo-keto-reductase. • Aldehydes reduction may be possible using continuous fermentation. In this experimental process, yeast is immobilized in specialized reactors under conditions, similar to CCF, which limit the cell metabolism. Adsorptive removal • Physisorption by hydrophobic resins and activated carbon • Chemisorption on aminated materials (materials containing an amine/amino-group) Separation by extraction • Supercritical carbon dioxide extraction • Extractant-impregnated resin technology Membrane based removal by pervaporation • Conversion into aroma-neutral or positive flavor compounds through oxidation, reduction or adduct formation Process during brewing to reduce aldehydes include: • Addition of binding compounds like amino acids, proteins, polyphenols like epicatechin, and sulphites like sulfur dioxide. • Thermal removal by stripping vacuum rectification or evaporation. For more insight into in aldehydes in non-alcoholic beer: Gernat, D.C., Brouwer, E. & Ottens, M. Aldehydes as Wort Off-Flavours in Alcohol-Free Beers—Origin and Control. Food Bioprocess Technol 13, 195–216 (2020). https://doi.org/10.1007/s11947-019-02374-z |
Higher Alcohols
Piornos et al (2022), in a literature review, provided the following insights into the influence of non-alcoholic beer brewing on higher alcohols [3].
Formation [3] Higher alcohols (as well as fatty acids, also known as carboxylic acids) are formed when yeasts degrade the nonpolar branched-chain amino acids: valine, leucine, and isoleucine. This process is composed of a multitude of reactions: Transamination reaction [24] • An amino acid’s amino group is transferred from one molecule to another [25]. In this case, the amino group is transferred from a branched-chain amino acid to α-ketoglutarate, thereby forming α-keto acid and glutamate. • The reaction is catalyzed by mitochondrial and cytosolic branched-chain amino acid aminotransferase (BCAATases) enzymes, encoded by the BAT1 and BAT2 genes. The anabolic pathway is a secondary formation mechanism of α-keto acid that occurs when fermentation media is low or absent of nitrogenous nutrients. In this process, yeasts synthesize the α-keto acid from the metabolism of glucose, via pyruvate. Ehrlich Pathway [26] • The α-keto acid is decarboxylated (removal of a carboxyl group) by decarboxylase enzymes which forms a fusel aldehyde (Strecker aldehyde). Alternatively, the Strecker aldehyde can be formed through the Maillard reaction [3]. • Then, depending on the redox (reduction-oxidation) status of the yeast: • If oxygen is present, the fusel aldehyde will be oxidized into a fusel acid/short-chain fatty acid. • If oxygen is not present, the fusel aldehyde will be reduced into a fusel alcohol by alcohol dehydrogenases. Important types in non-alcoholic beer: 3-methylbutanol, 2-methylbutanol, 2-phenylethanol, and 2-methylpropanol For more on higher alcohols: www.hawaiibevguide.com/ wine-aroma-compounds-pt-2.html#ehrlich-pathway Influenced By: Arrested fermentation and cold contact fermentation techniques, which use short time, low temperature (or both) to obtain a minimal reduction of aldehydes but also reduce the formation of higher alcohols. This, in turn, reduces ester formation, which exacerbates the undesirable aroma of Strecker aldehydes. Fermentation temperature, as it influences gene expression responsible for encoding a permease (membrane transport proteins that enable the diffusion in or out of the cell of a specific molecule) for amino acids into the yeast cell [3]. • This has a significant impact on the formation of higher alcohols via the Ehrlich pathway, and thus on the formation of esters. • Higher fermentation temperature improves the formation of higher alcohols, esters, and ethanol in beer. |
Esters [3]
|
Formation
• Esters = carboxylic acid + alcohol • Acetate esters = acetic acid + higher alcohol • Ethyl esters = carboxylic acid + ethanol • Acetic acid is produced by the oxidation of acetaldehyde Important types in non-alcoholic beer: Ethyl acetate, 3-methylbutyl acetate, 2- phenylethyl acetate. For more on ester formation: www.hawaiibevguide.com/wine-aroma-compounds-pt-2.html#esters Influenced By: • Higher alcohol and carboxylic acid production, as these are the precursors for esters. • Yeast type, as non-Saccharomyces cerevisiae yeast have been found to produce higher concentrations of 3-methylbutyl acetate, ethyl 2-methylpropanoate, and ethyl butanoate, which are the highly odor-active aroma compounds found in Bavarian wheat beers [27]. Sugar As modified fermentation techniques can create high amounts of unfermented sugar, this can result in beer with increased body and/or fullness compared to dealcoholized beer [1]. β-Damascenone (ketone) [3] Formation Precursors include: The carotenoid neoxanthin Process • Neoxanthin can be degraded by carotenoid cleavage dioxygenases into “grasshopper ketone” (named after its original isolation from the grasshopper, Romalea microptera) [28]. • Grasshopper ketone undergoes dehydration (the loss of water) and the transposition of oxygen from the C9 to C7 position to form β-damascenone. Impact on Beer • Contributes to the overall worty flavor of beer with a fruity, jam-like aroma. • Reportedly one of the compounds with the highest odor impact in alcohol-free beer brewed by cold contact fermentation (Piornos et al., 2020) and also in un-hopped wort and barley malt. Influenced by • Mashing and wort boiling, as its concentration changes in what is potentially a balance between formation and loss by evaporation, reaching a maximum at the end of wort boiling. • Increased temperature and acidic pH favor formation. • Hop type and quantity, as they can contribute glycosidically bound β-damascenone precursors like 3-hydroxy-β-damascone. |
Abhexone (5-ethyl-3-hydroxy-4-methyl-2(5H)-furanone) [3]
Impact on Beer Also known as “maple furanone,” it provides a curry, spicy aroma which contributes to the “hot honey” character of alcohol-free beer. Formation Proposed formation pathways • Aldol condensation of two molecules of α-ketobutyric acid originating from the degradation of threonine form a dimer. This, in turn, undergoes dehydration, cyclization, and decarboxylation reactions to further form abhexone and its keto-enol tautomer. The authors indicated that the predominant tautomer was abhexone in its enolic form. • Dimer: An oligomer (polymer with few repeating units) consisting of two monomers joined by bonds that can be either strong or weak, covalent or intermolecular [29] • Aldol condensation: A condensation reaction in which an enol or an enolate ion reacts with a carbonyl compound to form a β-hydroxyaldehyde or β-hydroxyketone, followed by dehydration to give a conjugated enone [30]. • Aldol condensation of α-ketobutyric acid and propanal. The formation of abhexone was observed after three days in wine-like matrices (13% ethanol, pH 1.98) even at room temperature (20 °C). Influence of Other Factors on Non-Alcoholic Beer While the production methodology which reduces or eliminates alcohol is influential on the final flavor of non-alcoholic beer, hopping methodologies may be even more influential. Ramsey et al (2021), in a study on understanding the sensory and physicochemical differences between commercially produced non-alcoholic lagers and their influence on consumer liking, notes: “Variation in sensory and physicochemical profiles of NABs may not only be due to the production methods used but also by other important factors including different starting raw materials (such as the addition of adjuncts including rye, wheat, rice or maize) or post processing methods (such as the use of additive flavour compounds, dry hopping or addition of liquid hop products post fermentation).” Of these methodologies, hopping techniques have been very significant to the flavor of non-alcoholic beer, because dry hopping and especially liquid hop products have been used to create IPA-style non-alcoholic beers, which help to mask overly intense hop aromas. Additionally, liquid hop products are microbially stable, thereby allowing their usage in conjunction with arrested fermentation techniques. For more insight into hopping methodologies and form factors, look out for a future Hawaii Beverage Guide article on the topic. |
Resources and Suggested Reading
1. Brányik, T., Silva, D. P., Baszczyňski, M., Lehnert, R., & e Silva, J. B. A. (2012). A review of methods of low alcohol and alcohol-free beer production. Journal of Food Engineering, 108(4), 493-506. Retrieved from:
https://pdfslide.net/documents/a-review-of-methods-of-low-alcohol-and-alcohol-free-beer-production.html?page=1
2. Montanari, L., Marconi, O., Mayer, H., & Fantozzi, P. (2009). Production of alcohol-free beer. In Beer in health and disease prevention (pp. 61-75). Academic Press. Retrieved from: www.researchgate.net/publication/286327980_Production_of_
alcohol-free_beer
3. Piornos, J. A., Koussissi, E., Balagiannis, D. P., Brouwer, E., & Parker, J. K. (2022). Alcohol-free and low-alcohol beers: Aroma chemistry and sensory characteristics. Comprehensive Reviews in Food Science and Food Safety. https://ift.onlinelibrary.wiley.com/doi/full/10.1111/1541-4337.13068
4. Thomas, K. (n.d.). Pitching. Craft Beer & Brewing. Retrieved February 3, 2023, from https://beerandbrewing
.com/dictionary/V04NmdFJJA/
5. Britannica, T. Editors of Encyclopaedia (2019, January 10). aliphatic compound. Encyclopedia Britannica. https://www.britannica.com/science/aliphatic-compound
6. Marchbanks, C. J. (n.d.). Original Gravity. Craft Beer & Brewing. Retrieved February 3, 2023, from
https://beerandbrewing.com/dictionary/c9EBwhgZpA/
7. Villa, K. (n.d.). High Gravity Brewing. Craft Beer & Brewing. Retrieved February 3, 2023,
from https://beerandbrewing.com/dictionary/4xtnZ4PwZ3/
8. Donaldson, B. (n.d.). Beer 101: Tips for Dry January. Lagunitas Brewing Company. Retrieved December 11, 2022,
from https://lagunitas.com/story/beereducation-dryjanuary/
9. Montanari, L., Floridi, S., Marconi, O., Tironzelli, M., & Fantozzi, P. (2005). Effect of mashing procedures on brewing. European Food Research and Technology, 221(1), 175-179. Retrieved from: https://www.researchgate.net/publication/225965293_Effect_of_mashing_procedures_on_brewing
10. Colby, C. (2006, December). Decoction Mashing Techniques. Brew Your Own. Retrieved February 3, 2023,
from https://byo.com/article/decoction-mashing-techniques/
11. National Center for Biotechnology Information (2023). PubChem Patent Summary for WO-2014135673-A2. Retrieved January 28, 2023 from. https://patents.google.com/patent/WO2014135673A2/en
12. Janish, S. (2021, September 8). Exploration of Post-Fermentation Hop Oil Extracts. ScottJanish.com. Retrieved February 13, 2023, from http://scottjanish.com/exploration-of-post-fermentation-hop-oil-extracts/
13. Carpenter, D. (2017, May 18). What Is the Difference between Ale and Lager? Craft Beer & Brewing. Retrieved February 13, 2023, from https://beerandbrewing.com/what-is-the-difference-between-ale-and-lager/
14. Navrátil, M., Dömény, Z., Šturdík, E., Šmogrovičová, D., & Gemeiner, P. (2002). Production of non-alcoholic beer using free and immobilized cells of Saccharomyces cerevisiae deficient in the tricarboxylic acid cycle. Biotechnology and applied biochemistry, 35(2), 133-140. https://www.academia.edu/18674730/Production_of_non_alcoholic_beer_using_free_and_
immobilized_cells_of_Saccharomyces_cerevisiae_deficient_in_the_tricarboxylic_acid_cycle
15. Strejc, J., Siříšťová, L., Karabín, M., Almeida e Silva, J.B. and Brányik, T. (2013), Production of alcohol-free beer with elevated amounts of flavouring compounds using lager yeast mutants. J. Inst. Brew., 119: 149-155. https://doi.org/10.1002/jib.72z
16. Piornos, J. A., Balagiannis, D. P., Methven, L., Koussissi, E., Brouwer, E., & Parker, J. K. (2020). Elucidating the odor-active aroma compounds in alcohol-free beer and their contribution to the worty flavor. Journal of agricultural and food chemistry, 68(37), 10088-10096. https://pubs.acs.org/doi/full/10.1021/acs.jafc.0c03902
Resources and Suggested Reading
17. White Labs Technical Team. (2021, January 28). Default Meta TitleTips for Brewing Non-Alcoholic and Low Alcohol Beer. White Labs. Retrieved February 28, 2023, from https://www.whitelabs.com/news-update-detail?id=44
18. Muller, C., Neves, L.E., Gomes, L., Guimarães, M. and Ghesti, G., 2019. Processes for alcohol-free beer production: a review. Food Science and Technology, 40, pp.273-281. https://doi.org/10.1590/fst.32318
19. Bellut, K., Michel, M., Hutzler, M., Zarnkow, M., Jacob, F., De Schutter, D. P., ... & Arendt, E. K. (2019). Investigation into the potential of Lachancea fermentati strain KBI 12.1 for low alcohol beer brewing. Journal of the American Society of Brewing Chemists, 77(3), 157-169. https://www.researchgae.net/publication/334554626_Investigation_into_
the_Potential_of_Lachancea_fermentati_Strain_KBI_121_for_Low_Alcohol_Beer_Brewing
20. Gernat, D.C., Brouwer, E. & Ottens, M. Aldehydes as Wort Off-Flavours in Alcohol-Free Beers—Origin and Control. Food Bioprocess Technol 13, 195–216 (2020). https://doi.org/10.1007/s11947-019-02374-z
21. Perpete, P., & Collin, S. (1999). Contribution of 3-methylthiopropionaldehyde to the worty flavor of alcohol-free beers. Journal of agricultural and food chemistry, 47(6), 2374-2378. https://doi.org/10.1021/jf9811323
22. Blanco, C. A., Andrés-Iglesias, C., & Montero, O. (2016). Low-alcohol beers: Flavor compounds, defects, and improvement strategies. Critical reviews in food science and nutrition, 56(8), 1379-1388. Retrieved from: https://www.researchgate.net/publication/264798799_Low-alcohol_Beers_Flavor_Compounds_Defects_and
_Improvement_Strategies
23. Perpète, P., & Collin, S. (1999). Fate of the worty flavours in a cold contact fermentation. Food Chemistry, 66(3), 359-363.
24. Styger, G., Prior, B., & Bauer, F. F. (2011). Wine flavor and aroma. Journal of Industrial Microbiology and Biotechnology, 38(9), 1145. Retrieved from: www.researchgate.net/publication/51518412_Wine_flavor_and_aroma
25. Wikipedia contributors. (2022, May 17). Transamination. In Wikipedia, The Free Encyclopedia. Retrieved 15:42, September 20, 2022, from https://en.wikipedia.org/w/index.php?title=Transamination&oldid=1088273136
26. Hazelwood, L. A., Daran, J. M., Van Maris, A. J., Pronk, J. T., & Dickinson, J. R. (2008). The Ehrlich pathway for fusel alcohol production: a century of research on Saccharomyces cerevisiae metabolism. Applied and environmental microbiology, 74(8), 2259-2266. https://doi.org/10.1128/AEM.02625-07
27. Bellut, K., & Arendt, E. K. (2019). Chance and challenge: Non-saccharomyces yeasts in nonalcoholic and low alcohol beer brewing–A review. Journal of the American society of brewing chemists, 77(2), 77-91. Retrieved from: https://www.researchgate.net/publication/331943504_Chance_and_Challenge_Non-_Saccharomyces_Yeasts_in_Nonalcoholic_and_Low_Alcohol_Beer_Brewing_-_A_Review
28. National Center for Biotechnology Information (2023). PubChem Compound Summary for CID 13922639, Grasshopper ketone. Retrieved February 3, 2023 from https://pubchem.ncbi.nlm.nih.gov/compound/Grasshopper-ketone.
29. Wikipedia contributors. (2023, January 30). Dimer (chemistry). In Wikipedia, The Free Encyclopedia. Retrieved 23:20, February 13, 2023, from https://en.wikipedia.org/w/index.php?title=Dimer_(chemistry)&oldid=1136519949
30. Aldol Condensation. (2023, January 22). https://chem.libretexts.org/@go/page/77955
31. Ramsey, I., Yang, Q., Fisk, I., & Ford, R. (2021). Understanding the sensory and physicochemical differences between commercially produced non-alcoholic lagers, and their influence on consumer liking. Food chemistry: X, 9, 100114.
https://www.sciencedirect.com/science/article/pii/S259015752100002X
PUBLISHED BY HAWAI'I BEVERAGE
MENU
|
HOME
|
SUBSCRIBE |
DIGITAL
|
BEVERAGE
|
NEws and
|
ABOUT |
CONTACT |
©2022 Hawaii Beverage Guide
Terms & Conditions
Terms & Conditions
- Subscribe
- Digital Edition
-
Beverage Guide
- Flavor and Cocktail Construction >
- Production Fundamentals >
- Non-Alcoholic Beverages >
-
Beer
>
- Cider >
- Sake
-
Spirits
>
-
Wine
>
- Kamaʻāina Wine Makers >
-
Winemaking
>
- A Guide to: High Sugar Residual Wines
- A Guide to: Post Fermentation Flavor Adjustments
- A Guide to: Post Fermentation Process: Stabilization
- A Guide to: Wine Prefermentation Practices
- A Guide to: Wine Microbes
- A Guide to: Wine Alcoholic Fermentation Physical Environment
- A Guide to: Wine Fermentation Chemical Environment
- A Guide to: Wine Bottling
- A Guide to: Wine Faults
- A Guide to: Wine Polyphenols
- A Guide to: Wine Aroma Compounds: Pt 1
- A Guide to: Wine Aroma Compounds: Pt 2
- A Guide to Viticulture
- Red and White Grape Aroma Compounds
- Wine Styles >
- Business Strategy
- News and Events
- About
- Production Fundamentals
- Flower Aroma Compounds